Page 5 - OpenAccess-April-2021
P. 5
TECHNICAL PAPER
however, ensures the production of hydroxyl ions on the surface low concentrations near the reinforcement as they are being
of the steel from the cathodic reaction (Equation 1). repelled away are unlikely to cause depassivation, so only
modest polarization is needed, typically an impressed current
-
½ O 2 + H 2 O + 2e → 2OH - (1)
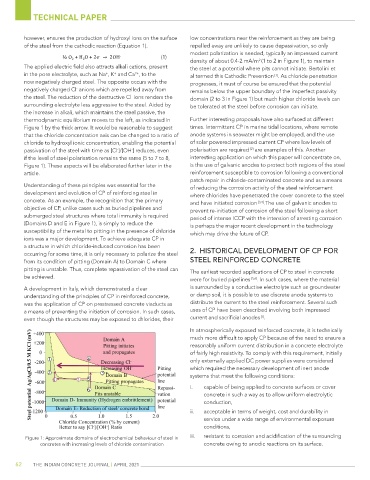
density of about 0.4-2 mA/m (1 to 2 in Figure 1), to maintain
2
The applied electric field also attracts alkali cations, present the steel at a potential where pits cannot initiate. Bertolini et
in the pore electrolyte, such as Na , K and Ca , to the al termed this Cathodic Prevention . As chloride penetration
+
2+
+
[1]
now negatively charged steel. The opposite occurs with the progresses, it must of course be ensured that the potential
negatively charged Cl anions which are repelled away from remains below the upper boundary of the imperfect passivity
-
the steel. The reduction of the destructive Cl ions renders the domain (2 to 3 in Figure 1) but much higher chloride levels can
-
surrounding electrolyte less aggressive to the steel. Aided by be tolerated at the steel before corrosion can initiate.
the increase in alkali, which maintains the steel passive, the
thermodynamic equilibrium moves to the left, as indicated in Further interesting proposals have also surfaced at different
Figure 1 by the thick arrow. It would be reasonable to suggest times. Intermittent CP in marine tidal locations, where remote
that the chloride concentration axis can be changed to a ratio of anode systems in seawater might be employed, and the use
chloride to hydroxyl ionic concentration, enabling the potential of solar powered impressed current CP where low levels of
[2]
passivation of the steel with time as [Cl ]/[OH ] reduces, even polarisation are required are examples of this. Another
-
-
if the level of steel polarisation remains the same (5 to 7 to 8, interesting application on which this paper will concentrate on,
Figure 1). These aspects will be elaborated further later in the is the use of galvanic anodes to protect both regions of the steel
article. reinforcement susceptible to corrosion following a conventional
patch repair in chloride-contaminated concrete and as a means
Understanding of these principles was essential for the of reducing the corrosion activity of the steel reinforcement
development and evolution of CP of reinforcing steel in where chlorides have penetrated the cover concrete to the steel
concrete. As an example, the recognition that the primary and have initiated corrosion [3,4] .The use of galvanic anodes to
objective of CP, unlike cases such as buried pipelines and prevent re-initiation of corrosion of the steel following a short
submerged steel structures where total immunity is required period of intense ICCP with the intension of arresting corrosion
(Domains D and E in Figure 1), is simply to reduce the is perhaps the major recent development in the technology
susceptibility of the metal to pitting in the presence of chloride which may drive the future of CP.
ions was a major development. To achieve adequate CP in
a structure in which chloride-induced corrosion has been
occurring for some time, it is only necessary to polarize the steel 2. HISTORICAL DEVELOPMENT OF CP FOR
from its condition of pitting (Domain A) to Domain C where STEEL REINFORCED CONCRETE
pitting is unstable. Thus, complete repassivation of the steel can The earliest recorded applications of CP to steel in concrete
be achieved. were for buried pipelines [5,6] . In such cases, where the material
A development in Italy, which demonstrated a clear is surrounded by a conductive electrolyte such as groundwater
understanding of the principles of CP in reinforced concrete, or damp soil, it is possible to use discrete anode systems to
was the application of CP on prestressed concrete viaducts as distribute the current to the steel reinforcement. Several such
a means of preventing the initiation of corrosion. In such cases, uses of CP have been described involving both impressed
[7]
even though the structures may be exposed to chlorides, their current and sacrificial anodes .
In atmospherically exposed reinforced concrete, it is technically
much more difficult to apply CP because of the need to ensure a
reasonably uniform current distribution in a concrete electrolyte
of fairly high resistivity. To comply with this requirement, initially
only externally applied DC power supplies were considered
which required the necessary development of inert anode
systems that meet the following conditions:
i. capable of being applied to concrete surfaces or cover
concrete in such a way as to allow uniform electrolytic
conduction,
ii. acceptable in terms of weight, cost and durability in
service under a wide range of environmental exposure
conditions,
Figure 1: Approximate domains of electrochemical behaviour of steel in iii. resistant to corrosion and acidification of the surrounding
concretes with increasing levels of chloride contamination concrete owing to anodic reactions on its surface.
62 THE INDIAN CONCRETE JOURNAL | APRIL 2021