Page 9 - OpenAccess-April-2021
P. 9
TECHNICAL PAPER
200 100
)
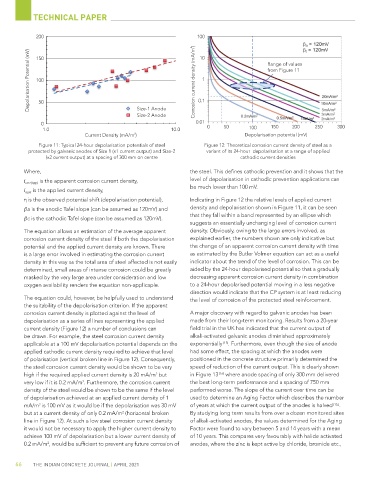
2 10 Range of values
Depolarisation Potential (mV) 100 Corrosion current density (mA/m 1 from Figure 11
150
50
Size-1 Anode
Size-2 Anode 0.1
0 0.01
1.0 10.0 0 50 100 150 200 250 300
p
Current Density (mA/m ) 2 Depolarisation otential (mV)
Figure 11: Typical 24-hour depolarisation potentials of steel Figure 12: Theoretical corrosion current density of steel as a
protected by galvanic anodes of Size 1 (x1 current output) and Size-2 variant of its 24-hour depolarisation at a range of applied
(x2 current output) at a spacing of 300 mm on centre cathodic current densities
Where, the steel. This defines cathodic prevention and it shows that the
i corr(app) is the apparent corrosion current density, level of depolarisation in cathodic prevention applications can
be much lower than 100 mV.
i appl is the applied current density,
η is the observed potential shift (depolarisation potential), Indicating in Figure 12 the relative levels of applied current
a is the anodic Tafel slope (can be assumed as 120mV) and density and depolarisation shown in Figure 11, it can be seen
that they fall within a band represented by an ellipse which
c is the cathodic Tafel slope (can be assumed as 120mV).
suggests an essentially unchanging level of corrosion current
The equation allows an estimation of the average apparent density. Obviously, owing to the large errors involved, as
corrosion current density of the steel if both the depolarisation explained earlier, the numbers shown are only indicative but
potential and the applied current density are known. There the change of an apparent corrosion current density with time
is a large error involved in estimating the corrosion current as estimated by the Butler Volmer equation can act as a useful
density in this way as the total area of steel affected is not easily indicator about the trend of the level of corrosion. This can be
determined, small areas of intense corrosion could be greatly aided by the 24-hour depolarised potential so that a gradually
masked by the very large area under consideration and low decreasing apparent corrosion current density in combination
oxygen availability renders the equation non-applicaple. to a 24-hour depolarised potential moving in a less negative
direction would indicate that the CP system is at least reducing
The equation could, however, be helpfully used to understand the level of corrosion of the protected steel reinforcement.
the suitability of the depolarisation criterion. If the apparent
corrosion current density is plotted against the level of A major discovery with regard to galvanic anodes has been
depolarisation as a series of lines representing the applied made from their long-term monitoring. Results from a 20-year
current density (Figure 12) a number of conclusions can field trial in the UK has indicated that the current output of
be drawn. For example, the steel corrosion current density alkali-activated galvanic anodes diminished approximately
applicable at a 100 mV depolarisation potential depends on the exponentially [15] . Furthermore, even though the size of anode
applied cathodic current density required to achieve that level had some effect, the spacing at which the anodes were
of polarisation (vertical broken line in Figure 12). Consequently, positioned in the concrete structure primarily determined the
the steel corrosion current density would be shown to be very speed of reduction of the current output. This is clearly shown
high if the required applied current density is 20 mA/m but in Figure 13 [16] where anode spacing of only 300 mm delivered
2
very low if it is 0.2 mA/m . Furthermore, the corrosion current the best long-term performance and a spacing of 750 mm
2
density of the steel would be shown to be the same if the level performed worse. The slope of the current over time can be
of depolarisation achieved at an applied current density of 1 used to determine an Aging Factor which describes the number
2
mA/m is 100 mV as it would be if the depolarisation was 30 mV of years at which the current output of the anodes is halved [15,] .
but at a current density of only 0.2 mA/m (horizontal broken By studying long term results from over a dozen monitored sites
2
line in Figure 12). At such a low steel corrosion current density of alkali-activated anodes, the values determined for the Aging
it would not be necessary to apply the higher current density to Factor were found to vary between 5 and 14 years with a mean
achieve 100 mV of depolarisation but a lower current density of of 10 years. This compares very favourably with halide activated
0.2 mA/m , would be sufficient to prevent any future corrosion of anodes, where the zinc is kept active by chloride, bromide etc.,
2
66 THE INDIAN CONCRETE JOURNAL | APRIL 2021