Page 6 - July-Open-Access-2020
P. 6
TECHNICAL PAPER
4.2 Water quantity
The water/cement ratio is one of the most important parameters
of Portland cement concretes and this ratio is limited for all
the exposure classes of standard EN 206. In the case of alkali-
activated materials, the water seems to have quite different
functions depending on the precursor/activator couple used.
In the case of slag, the reaction products are essentially C-a-
s-H with a low Ca/si ratio, so the water is partly contained
in hydrates, as in Portland cements. sulfate activation also
produces ettringite, which includes water in its structure.
On the other hand, aluminosilicates (MK and class F fly ash)
attacked by an alkali-silicate form a three-dimensional network,
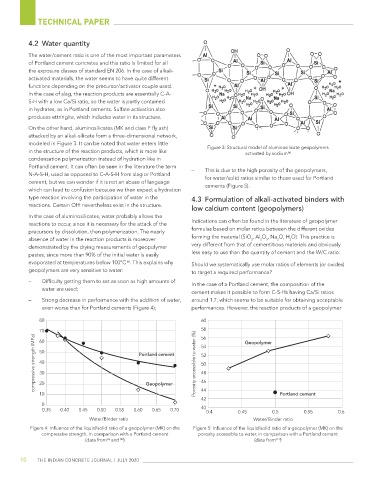
modeled in Figure 3. It can be noted that water enters little
in the structure of the reaction products, which is more like Figure 3: structural model of aluminosilicate geopolymers
activated by sodium
[4]
condensation polymerization instead of hydration like in
Portland cement. It can often be seen in the literature the term – This is due to the high porosity of the geopolymers,
N-a-s-H, used as opposed to C-a-s-H from slag or Portland for water/solid ratios similar to those used for Portland
cement, but we can wonder if it is not an abuse of language
which can lead to confusion because we then expect a hydration cements (Figure 5).
type reaction involving the participation of water in the 4.3 Formulation of alkali-activated binders with
reactions. Certain OH nevertheless exist in the structure.
-
low calcium content (geopolymers)
In the case of aluminosilicates, water probably allows the
reactions to occur, since it is necessary for the attack of the Indications can often be found in the literature of geopolymer
precursors by dissolution, then polymerization. The nearly formulas based on molar ratios between the different oxides
absence of water in the reaction products is moreover forming the material (siO , al O , Na O, H O). This practice is
2
2
3
2
2
demonstrated by the drying measurements of geopolymer very different from that of cementitious materials and obviously
pastes, since more than 90% of the initial water is easily less easy to use than the quantity of cement and the W/C ratio.
evaporated at temperatures below 100°C . This explains why should we systematically use molar ratios of elements (or oxides)
[6]
geopolymers are very sensitive to water: to target a required performance?
– Difficulty getting them to set as soon as high amounts of In the case of a Portland cement, the composition of the
water are used;
cement makes it possible to form C-s-Hs having Ca/si ratios
– strong decrease in performance with the addition of water, around 1.7, which seems to be suitable for obtaining acceptable
even worse than for Portland cements (Figure 4); performances. However, the reaction products of a geopolymer
Figure 4: Influence of the liquid/solid ratio of a geopolymer (MK) on the Figure 5: Influence of the liquid/solid ratio of a geopolymer (MK) on the
compressive strength, in comparison with a Portland cement porosity accessible to water, in comparison with a Portland cement
(data from and ) (data from )
[11]
[6]
[10]
10 The IndIan ConCreTe Journal | JulY 2020