Page 30 - openaccess
P. 30
TECHNICAL PAPER
7.2 Geo-chemical investigations anions concentration (for example; 24 (O) for hornblende, 18
(O) for clinohumite) proposed by Deer et al. . Upon calculating
[7]
The study of geochemistry of minerals is essential to identify equivalent anion concentration from the normalized oxides
the origin, mineral proportions, and other related engineering compositions, the mineral assemblages are calculated based
properties of aggregates. For such analyses, suitable geological on geochemical assumption pertaining to the exposures.
procedures can be employed. With regard to mineral Moreover, the norm aids in profiling the cationic exchanges due
assemblage, and alterations, Rittman’s norm [90] calculations to any exposure by supplementing the subtraction or addition
may be used [21] . It is a geochemical procedure in which, oxide plots. A comprehensive Rittmann’s norm analysis performed by
compositions are re-calculated into equivalent number of Haneefa et al. [21] is provided in Table 8. This norm differentiates
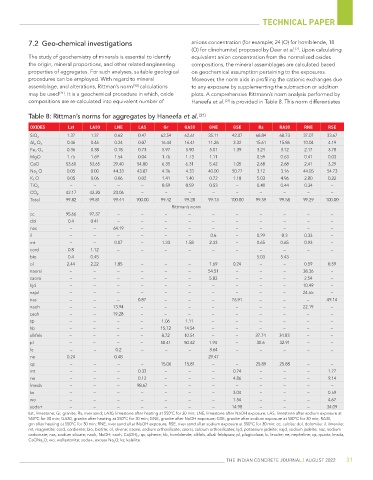
Table 8: Rittman’s norms for aggregates by Haneefa et al. [21]
OXIDES Lst LA30 LNE LAS Gr GA30 GNE GSE Rs RA30 RNE RSE
1.37 1.37 0.62 0.47 62.54 62.61 35.11 42.07 68.84 68.73 37.07 33.67
SiO 2
0.46 0.46 0.24 0.07 16.44 16.41 11.26 3.32 15.61 15.86 10.04 4.19
Al 2 O 3
0.36 0.38 0.18 0.73 5.97 5.90 5.01 1.39 3.24 3.12 2.17 3.78
Fe 2 O 3
MgO 1.75 1.69 1.54 0.04 1.76 1.73 1.11 0.59 0.63 0.41 0.03
CaO 53.60 53.65 29.40 54.80 6.35 6.31 5.42 1.05 2.68 2.68 2.41 3.29
Na 2 O 0.05 0.00 44.33 43.87 4.36 4.33 40.00 50.77 3.12 3.16 44.05 54.73
K 2 O 0.05 0.06 0.06 0.02 1.41 1.40 0.72 1.18 5.03 4.96 2.80 0.22
– – – – 0.59 0.59 0.53 – 0.48 0.44 0.34 –
TiO 2
42.17 42.20 23.06 – – – – – – – – –
CO 2
Total 99.82 99.81 99.44 100.00 99.42 99.28 99.13 100.00 99.59 99.58 99.29 100.00
Rittman’s norm
cc 95.66 97.37 – – – – – – – – – –
dol 0.4 0.41 – – – – – – – – – –
nac – – 64.19 – – – – – – – – –
il – – – – – – 0.6 – 0.29 0.3 0.33 –
mt – – 0.07 – 1.33 1.58 2.33 – 0.65 0.65 0.93 –
cord 0.8 1.12 – – – – – – – – – –
bio 0.4 0.45 – – – – – – 5.03 5.43 – –
ol 2.44 2.22 1.85 – – – 1.69 0.24 – – 0.59 0.59
naorsi – – – – – – 54.51 – – – 38.36 –
caorsi – – – – – – 5.83 – – – 2.54 –
kjd – – – – – – – – – – 10.49 –
najd – – – – – – – – – – 24.55 –
nas – – – 0.87 – – – 76.91 – – – 49.14
naoh – – 13.94 – – – – – – – 22.19 –
caoh – – 19.28 – – – – – – – – –
sp – – – – 1.06 1.11 – – – – – –
hb – – – – 15.72 14.54 – – – – – –
alkfels – – – – 8.72 10.54 – – 37.74 34.83 – –
pl – – – – 58.41 50.42 1.94 – 30.6 32.91 – –
lc – – 0.2 – – – 3.64 – – – – –
ne 0.24 – 0.48 – – – 29.47 – – – – –
qz – – – – 15.06 15.81 – – 25.89 25.88 – –
mt – – – 0.33 – – – 0.74 – – – 1.77
ne – – – 0.12 – – – 4.86 – – – 9.14
lmsda – – – 98.67 – – – – – – – –
ks – – – – – – – 3.04 – – – 0.59
wo – – – – – – – 1.54 – – – 4.67
soda+ – – – – – – – 14.98 – – – 34.09
Lst, limestone; Gr, granite; Rs, river sand; LA30, limestone after heating at 550°C for 30 min; LNE, limestone after NaOH exposure; LAS, limestone after sodium exposure at
550°C for 30 min; GA30, granite after heating at 550°C for 30 min; GNE, granite after NaOH exposure; GSE, granite after sodium exposure at 550°C for 30 min; RA30,
grn after heating at 550°C for 30 min; RNE, river sand after NaOH exposure, RSE, river sand after sodium exposure at 550°C for 30 min; cc, calcite; dol, dolomite; il, ilmenite;
mt, magnetite; cord, cordierite; bio, biotite; ol, olivine; naorsi, sodium orthosilicate; caorsi, calcium orthosilicates; kjd, potassium jadeite; najd, sodium jadeite; nac, sodium
carbonate; nas, sodium silicate; naoh, NaOH; caoh, Ca(OH) 2 ; sp, sphene; hb, hornblende; alkfels, alkali feldspars; pl, plagioclase; lc, leucite; ne, nepheline; qz, quartz; lmsda,
CaONa 2 O; wo; wollastonite; soda+, excess Na 2 O; ks; kalsilite
THE INDIAN CONCRETE JOURNAL | AUGUST 2022 31