Page 20 - openaccess
P. 20
TECHNICAL PAPER
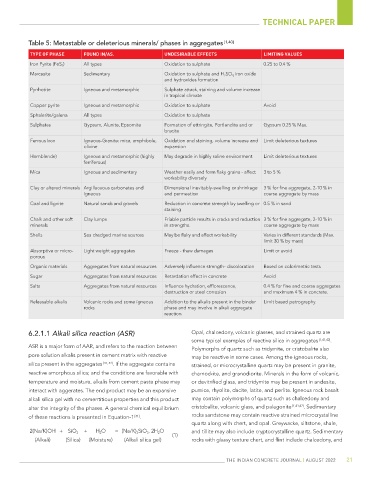
Table 5: Metastable or deleterious minerals/ phases in aggregates [1,40]
TYPE OF PHASE FOUND IN/AS. UNDESIRABLE EFFECTS LIMITING VALUES
Iron Pyrite (FeS 2 ) All types Oxidation to sulphate 0.25 to 0.4 %
Marcasite Sedimentary Oxidation to sulphate and H 2 SO 4, iron oxide
and hydroxides formation
Pyrrhotite Igneous and metamorphic Sulphate attack, staining and volume increase
in tropical climate
Copper pyrite Igneous and metamorphic Oxidation to sulphate Avoid
Sphalerite/galena All types Oxidation to sulphate
Sulphates Gypsum, Alunite, Epsomite Formation of ettringite, Portlandite and or Gypsum 0.25 % Max.
brucite
Ferrous Iron Igneous-Granite: mica, amphibole, Oxidation and staining, volume increase and Limit deleterious textures
olivine expansion
Hornblende) Igneous and metamorphic (highly May degrade in highly saline environment Limit deleterious textures
ferriferous)
Mica Igneous and sedimentary Weather easily and form flaky grains - affect 3 to 5 %
workability diversely
Clay or altered minerals Argillaceous carbonates and Dimensional inevitably-swelling or shrinkage 3 % for fine aggregate, 2-10 % in
igneous and permeation coarse aggregate by mass
Coal and lignite Natural sands and gravels Reduction in concrete strength by swelling or 0.5 % in sand
staining
Chalk and other soft Clay lumps Friable particle results in cracks and reduction 3 % for fine aggregate, 2-10 % in
minerals in strengths. coarse aggregate by mass
Shells Sea dredged marine sources May be flaky and affect workability Varies in different standards (Max.
limit 30 % by mass)
Absorptive or micro- Light weight aggregates Freeze - thaw damages Limit or avoid
porous
Organic materials Aggregates from natural resources Adversely influence strength- discoloration Based on colorimetric tests
Sugar Aggregates from natural resources Retardation effect in concrete Avoid
Salts Aggregates from natural resources Influence hydration, efflorescence, 0.4 % for fine and coarse aggregates
destruction or steel corrosion and maximum 4 % in concrete.
Releasable alkalis Volcanic rocks and some igneous Addition to the alkalis present in the binder Limit based petrography.
rocks phase and may involve in alkali aggregate
reaction
6.2.1.1 Alkali silica reaction (ASR) Opal, chalcedony, volcanic glasses, and strained quartz are
some typical examples of reactive silica in aggregates [1,41,42] .
ASR is a major form of AAR, and refers to the reaction between
Polymorphs of quartz such as tridymite, or cristobalite also
pore solution alkalis present in cement matrix with reactive may be reactive in some cases. Among the igneous rocks,
silica present in the aggregates [41, 42] . If the aggregate contains strained, or microcrystalline quartz may be present in granite,
reactive amorphous silica; and the conditions are favorable with charnockite, and granodiorite. Minerals in the form of volcanic,
temperature and moisture, alkalis from cement paste phase may or devitrified glass, and tridymite may be present in andesite,
interact with aggerates. The end product may be an expansive pumice, rhyolite, dacite, latite, and perlite. Igneous rock basalt
alkali silica gel with no cementitious properties and this product may contain polymorphs of quartz such as chalcedony and
alter the integrity of the phases. A general chemical equilibrium cristobalite, volcanic glass, and palagonite [1,41,42] . Sedimentary
of these reactions is presented in Equation-1 [41] . rocks sandstone may contain reactive strained microcrystalline
quartz along with chert, and opal. Greywacke, siltstone, shale,
2(Na/K)OH + SiO 2 + H 2 O = (Na/K) 2 SiO 3 . 2H 2 O and tillite may also include cryptocrystalline quartz. Sedimentary
(1)
(Alkali) (Silica) (Moisture) (Alkali silica gel) rocks with glassy texture chert, and flint include chalcedony, and
THE INDIAN CONCRETE JOURNAL | AUGUST 2022 21