Page 24 - openaccess
P. 24
TECHNICAL PAPER
Figure 16: SEM-BSE image of (a) OPC concrete; and (b) Geopolymer; with limestone aggregate
after exposure to hot liquid sodium, Haneefa et al. (2013) [64, 68]
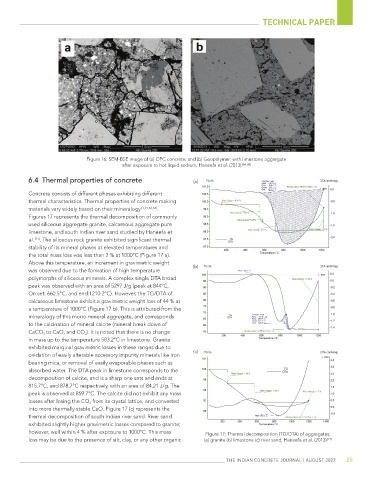
6.4 Thermal properties of concrete (a)
Concrete consists of different phases exhibiting different
thermal characteristics. Thermal properties of concrete making
materials vary widely based on their mineralogy [1,21,63,64] .
Figures 17 represents the thermal decomposition of commonly
used siliceous aggregate granite, calcareous aggregate pure
limestone, and south Indian river sand studied by Haneefa et
al. [21] . The siliceous rock granite exhibited significant thermal
stability of its mineral phases at elevated temperatures and
the total mass loss was less than 2 % at 1000°C (Figure 17 a).
Above this temperature, an increment in gravimetric weight
was observed due to the formation of high temperature (b)
polymorphs of siliceous minerals. A complex single DTA broad
peak was observed with an area of 5297 J/g (peak at 844°C,
On set: 660.5°C, and end:1210.2°C). However, the TG/DTA of
calcareous limestone exhibit a gravimetric weight loss of 44 % at
a temperature of 1000°C (Figure 17 b). This is attributed from the
mineralogy of this mono-mineral aggregate, and corresponds
to the calcination of mineral calcite (mineral break down of
CaCO 3 to CaO, and CO 2 ). It is noted that there is no change
in mass up to the temperature 583.2°C in limestone. Granite
exhibited marginal gravimetric losses in these ranges due to
oxidation of easily alterable accessory impurity minerals like iron (c)
bearing mica, or removal of easily evaporable phases such as
absorbed water. The DTA peak in limestone corresponds to the
decomposition of calcite, and is a sharp one sets and ends at
815.7°C, and 878.7°C respectively, with an area of 84.21 J/g. The
peak is observed at 859.7°C. The calcite did not exhibit any mass
losses after losing the CO 2 from its crystal lattice, and converted
into more thermally stable CaO. Figure 17 (c) represents the
thermal decomposition of south Indian river sand. River sand
exhibited slightly higher gravimetric losses compared to granite;
however, well within 4 % after exposure to 1000°C. This mass Figure 17: Thermal decomposition (TG/DTA) of aggregates;
loss may be due to the presence of silt, clay, or any other organic (a) granite (b) limestone (c) river sand, Haneefa et al. (2013) [21]
THE INDIAN CONCRETE JOURNAL | AUGUST 2022 25